Retinal vein occlusion (RVO) is a leading cause of vision loss, impacting millions globally. Treatments have evolved significantly, with anti-VEGF therapies revolutionizing care since their introduction in 2010. Faricimab, approved in late 2023, is the first dual-target therapy addressing both VEGF-A and Ang-2, offering longer-lasting control and fewer injections. Clinical trials show it improves vision and reduces retinal swelling more effectively than earlier treatments like aflibercept and ranibizumab. Emerging options, including gene therapies and extended-release implants, aim to further reduce treatment burdens. Early diagnosis and consistent care remain critical for preserving vision.
Justin H. Townsend, M.D. discusses retinal vein occlusion
sbb-itb-246add9
Faricimab (Vabysmo): A Dual-Target Treatment for RVO
Faricimab represents a new chapter in treating retinal vein occlusion (RVO). Unlike earlier therapies that solely block VEGF-A, this is the first bispecific antibody designed to target two pathways at once: VEGF-A and angiopoietin-2 (Ang-2). Approved by the FDA on October 26, 2023, for macular edema caused by RVO, it offers a more comprehensive approach to managing the condition.
This dual mechanism addresses two key issues in RVO. VEGF-A is responsible for fluid leakage and the growth of abnormal blood vessels, while elevated Ang-2 levels destabilize blood vessels and intensify inflammation. By simultaneously targeting both, faricimab stabilizes blood vessels more effectively than single-target therapies, providing longer-lasting control over retinal swelling. Clinical trials have further backed these benefits.
Clinical Trial Results
Faricimab’s effectiveness has been demonstrated in two Phase III trials: BALATON for branch RVO and COMINO for central RVO. Both studies reported significant vision improvements, with patients gaining an average of 16.9 letters on the eye chart by week 24. Additionally, it showed remarkable reductions in retinal swelling. Central subfield thickness decreased by 311.4 µm in BALATON and 461.6 µm in COMINO.
One of faricimab’s standout features is its ability to resolve macular edema more effectively than previous treatments. In the BALATON trial, 34% of patients achieved complete macular leakage resolution compared to 21% of those treated with aflibercept. In COMINO, the results were even more striking: 44% versus 30%. Long-term data at 72 weeks confirmed these benefits, with vision gains reaching 18.1 letters in BALATON and maintaining 16.9 letters in COMINO. For a chronic condition like RVO, these results are a game-changer.
Extended Dosing Benefits
Managing RVO has traditionally been demanding, requiring frequent injections every one to two months. Faricimab introduces a more flexible approach. Using a personalized "treat-and-extend" method, doctors can adjust injection schedules based on individual patient responses. This not only enhances vision outcomes but also reduces the treatment burden.
By week 72, nearly 60% of patients in the BALATON trial and up to 48% in COMINO were receiving injections every three to four months. Fewer treatments significantly improve the quality of life for patients.
"Nearly 60% of people receiving Vabysmo in BALATON and up to 48% of people in COMINO were able to extend their treatment intervals to 3 or 4 months apart. This is important, as there is a significant treatment burden associated with frequent eye injections." – Carl Danzig, MD, Director of Retina Clinical Research at Rand Eye Institute
Standard Anti-VEGF Therapies: Aflibercept and Ranibizumab
Even with faricimab offering a dual-pathway approach, it’s important to understand the role of standard anti-VEGF therapies in managing RVO (retinal vein occlusion). Aflibercept (Eylea) and ranibizumab (Lucentis) have been the primary choices for treating RVO-related macular edema for years. Both focus on blocking VEGF-A, a protein that promotes abnormal blood vessel growth. However, their mechanisms differ: aflibercept is a fusion protein that acts as a decoy receptor for VEGF-A, VEGF-B, and PlGF, while ranibizumab is a monoclonal antibody fragment targeting VEGF-A alone. Unlike faricimab, which addresses two pathways, these therapies zero in on VEGF-A inhibition.
Clinical trials highlight their effectiveness. Aflibercept demonstrated strong results in the COPERNICUS and GALILEO trials for central RVO, while ranibizumab showed similar outcomes in the BRAVO trial for branch RVO and the CRUISE trial for central RVO. One key distinction is that aflibercept trials included a higher percentage (8–20%) of patients with ischemic RVO, reflecting more complex cases compared to ranibizumab studies. Both treatments share a similar safety profile, with severe complications like endophthalmitis or retinal detachment affecting fewer than 1% of patients.
Efficacy and Safety Data
Both aflibercept and ranibizumab consistently deliver substantial visual improvements. In pivotal trials, patients typically gained 17 to 19 letters on eye charts within 24 weeks. Real-world data from England, which tracked 7,079 eyes over a decade (2013–2023), showed branch RVO patients achieving an average improvement of 11.2 letters at six months, while central RVO patients gained 11.5 letters during the same period. Aflibercept has shown a slight advantage in some cases. For example, a 12-month study on central RVO reported that aflibercept-treated patients achieved an average vision gain of +16.6 letters, compared to +9.8 letters for ranibizumab. Additionally, aflibercept reduced central subfield thickness by 304µm, outperforming ranibizumab’s 252µm reduction.
"Aflibercept performed slightly better than ranibizumab for treating eyes with CRVO in this study. Significant differences in the molecular structure and mode of action of the drugs we studied may be the reason for the better outcomes we found with aflibercept for CRVO." – Niedzwiecki M, et al.
Maintenance and Dosing Schedules
After initial improvements, treatment typically involves monthly injections – 0.5 mg for ranibizumab and 2 mg for aflibercept. Following a loading phase of at least three injections within the first 120 days, many patients switch to a PRN (pro re nata, or as-needed) dosing schedule for long-term maintenance. This approach reduces the number of injections but requires close monitoring. While early gains are significant, they tend to decline over time as injection frequency decreases. For example, by 60 months, branch RVO patients retained an average improvement of +8.3 letters, down from the initial 11.2 letters, with annual injections dropping from 7.0 in the first year to just 2.0 by the fifth year.
The newer aflibercept 8 mg formulation (Eylea HD), approved by the FDA in November 2025, offers extended dosing intervals. After the loading phase, patients can receive injections every eight weeks, potentially halving the number of treatments required.
Steroid Implants for Macular Edema
When anti-VEGF treatments fail to work for RVO (retinal vein occlusion), steroid implants can be a reliable alternative. These implants tackle inflammation by reducing cytokines like IL-6 and IL-8, stabilizing the blood-retinal barrier, and even blocking VEGF production. In cases of central RVO, steroids may also help reduce optic nerve head swelling, which can improve blood flow. Typically, these implants are considered a second-line option for patients who don’t respond to anti-VEGF treatments or those with vitrectomized eyes. By focusing on inflammation, steroid implants complement earlier anti-VEGF and faricimab therapies, offering a different approach for more complex RVO cases.
Dexamethasone Implants
The dexamethasone implant, known as Ozurdex, contains 0.7 mg of biodegradable medication that dissolves into water and carbon dioxide over time. The GENEVA study, which included 1,267 participants, found that Ozurdex helped patients gain 15 letters of vision improvement between days 30 and 90. In real-world scenarios, patients typically see an improvement of +7.8 letters within 12 weeks of treatment. The implant is most effective around two months after injection and usually requires retreatment by four months. However, retreating every three to five months has shown better results compared to the GENEVA study’s six-month schedule.
"The dexamethasone implant… usually provides clinically meaningful effects for 3 to 4 months based on our experience with refractory edema." – Yoshihiro Yonekawa, MD, and Jeremy D. Wolfe, MD, Associated Retinal Consultants
Side effects include increased intraocular pressure (IOP), which peaks around six to eight weeks and typically returns to normal levels after three to four months. Interestingly, 99% of patients who experience ocular hypertension due to Ozurdex do so within the first four injections. If no pressure spike occurs during this period, future doses are unlikely to cause issues. The risk of needing glaucoma surgery is low, occurring in only 0.6% of patients during large clinical trials. However, cataract progression is a common concern, with over 50% of patients experiencing lens changes during long-term follow-up (average 50.5 months).
Fluocinolone Acetonide Implants
Unlike dexamethasone, fluocinolone acetonide implants (Iluvien) are designed for extended release. This non-biodegradable implant delivers 0.19 mg of medication over an impressive 36 months from a single injection. Though primarily used off-label for RVO, it’s a great option for patients looking to reduce the frequency of injections. However, the longer duration comes with higher risks. The chance of requiring glaucoma surgery is 4.8%, significantly higher than the 0.6% risk with Ozurdex. This is likely because fluocinolone, being less water-soluble, may accumulate in the eye’s drainage system, leading to sustained IOP increases. For this reason, the FDA advises using Iluvien only in patients who have previously tolerated steroids without major pressure-related complications.
| Feature | Dexamethasone (Ozurdex) | Fluocinolone Acetonide (Iluvien) |
|---|---|---|
| Dose | 0.7 mg | 0.19 mg |
| Implant Type | Biodegradable | Non-biodegradable |
| Duration | 3–5 months (real-world) | Up to 36 months |
| IOP Surgery Risk | 0.6% | 4.8% |
| Primary Use | FDA-approved for RVO | Off-label for RVO |
New and Future Therapies for RVO
Researchers are exploring gene therapies and next-generation biologics to reduce the frequency of injections and ease the treatment process for retinal vein occlusion (RVO). These advancements build on the foundation of existing anti-VEGF and steroid implant therapies.
Gene Therapy Options
Gene therapy is pushing boundaries, aiming to enable retinal cells to produce anti-VEGF proteins continuously after a single injection. This method eliminates the "peak and trough" drug levels seen with traditional injections by maintaining consistent therapeutic concentrations. These therapies rely on viral vectors to deliver genetic material directly into the eye.
Ixoberogene soroparvovec (Ixo-vec / ADVM-022) utilizes an AAV.7m8 vector to deliver a transgene encoding aflibercept. Results from the Phase 2 LUNA trial showed that 69% of patients in the 2E11 dose group avoided additional injections for a year, while 90% required no more than two supplemental injections. In the Phase 1 OPTIC trial for wet AMD, patients experienced an 87% reduction in annual injection burden over four years.
"Gene therapy offers the potential to fully eliminate or reduce the requirement for ongoing IVT injections… achieving sustained therapeutic concentrations and the potential for lifelong clinical benefit." – Glenn Yiu, MD, PhD, Professor of Ophthalmology, UC Davis
RGX-314 (ABBV-RGX-314) uses an AAV8 vector to express a monoclonal antibody fragment similar to ranibizumab. It can be administered via subretinal injection, which requires surgery, or through suprachoroidal delivery, an in-office procedure. The Phase 2 AAVIATE trial revealed that patients receiving the highest dose via suprachoroidal delivery experienced an 80% reduction in annual injections.
4D-150 employs an R100 vector to produce aflibercept and a VEGF-C inhibitory miRNA, targeting multiple VEGF isoforms (A, B, C) along with placental growth factor. In the Phase 1/2 PRISM trial, patients saw a 97.5% reduction in injection rates over 36 weeks.
| Therapy | Vector | Therapeutic Protein | Delivery Route |
|---|---|---|---|
| Ixo-vec (ADVM-022) | AAV.7m8 | Aflibercept | Intravitreal |
| RGX-314 | AAV8 | Ranibizumab fragment | Subretinal / Suprachoroidal |
| 4D-150 | R100 | Aflibercept + VEGF-C miRNA | Intravitreal |
Novel Biologics and Additional Treatments
New biologics aim to improve treatment outcomes while reducing the need for frequent injections through innovative delivery methods.
Tarcocimab (KSI-301) is an antibody biopolymer conjugate combining a monoclonal anti-VEGF antibody with a high molecular weight polymer to extend its activity in the eye. In the Phase 3 BEACON trial, which included 561 patients with macular edema due to RVO, tarcocimab achieved its primary endpoint. Patients treated with tarcocimab received an average of 3.9 injections over 24 weeks compared to 5.8 injections in the aflibercept group, while maintaining similar visual acuity improvements. Notably, 46% of tarcocimab-treated patients gained 15 or more ETDRS letters.
Aflibercept 8 mg, a high-dose version of the standard therapy, is being tested in the Phase 3 QUASAR trial to extend treatment intervals beyond the usual 4–8 weeks. Additionally, the Port Delivery System (PDS), marketed as Susvimo, offers continuous delivery of ranibizumab via a refillable implant. After updates to its design and needle, it was reintroduced in July 2024, though trials specific to RVO are still pending.
Comparing Anti-VEGF Therapies: Efficacy, Safety, and Dosing
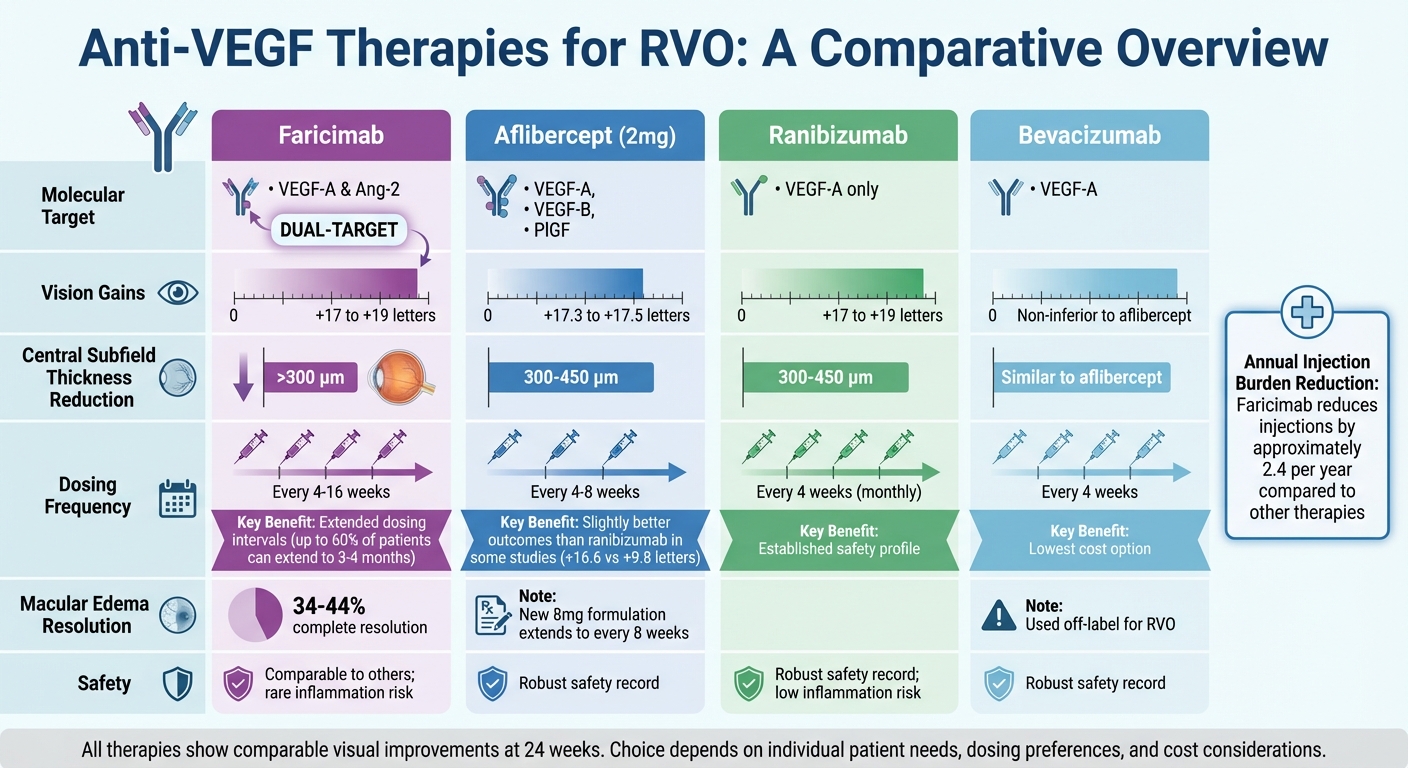
Comparison of Anti-VEGF Therapies for Retinal Vein Occlusion Treatment
Choosing the right anti-VEGF therapy means weighing factors like vision improvement, dosing schedules, and side effects. Faricimab is unique as the first bispecific antibody targeting both VEGF-A and angiopoietin-2 (Ang-2). This dual-target approach has shown better anatomical outcomes compared to aflibercept, with higher rates of complete macular leakage resolution in both branch and central RVO trials. While visual gains are similar to standard treatments, faricimab’s mechanism offers added benefits for retinal health.
At 24 weeks, all anti-VEGF agents deliver comparable visual improvements, typically ranging from +17 to +19 letters, and reduce central subfield thickness by 300–450 μm. However, dosing schedules differ significantly. Faricimab can extend dosing intervals up to 16 weeks for many patients, while aflibercept generally reaches up to 8 weeks, and ranibizumab often requires monthly injections. A meta-analysis highlights that faricimab reduces the annual injection burden by around 2.4 injections compared to other therapies.
"Faricimab’s dual mechanism of action, which involves inhibition of both VEGF and angiopoietin-2, may offer benefits beyond VEGF inhibition alone, addressing aspects of retinal vascular pathology more thoroughly and improving anatomical outcomes in patients with RVO." – Frontiers in Pharmacology
While all therapies provide similar vision improvements, their safety profiles and durability vary. Most anti-VEGF agents share comparable safety records, though real-world data suggests faricimab may have a slightly higher risk of intraocular inflammation compared to ranibizumab and bevacizumab. Bevacizumab, despite being used off-label, offers efficacy similar to aflibercept at a much lower cost. For patients unresponsive to standard treatments, switching to faricimab has shown promise. A study from Ulm University Hospital in Germany reported that patients with persistent fluid, despite a median of 23 prior injections, experienced a reduction in central subfield thickness from 291 μm to 268 μm after switching. Treatment intervals also extended from 4.6 weeks to 7.3 weeks.
Comparison Table of Anti-VEGF Agents
| Agent | Molecular Target(s) | BCVA Gains (Letters) | CST Reduction | Dosing Frequency | Safety Profile |
|---|---|---|---|---|---|
| Faricimab | VEGF-A & Ang-2 | +17 to +19 | >300 μm | Every 4–16 weeks | Comparable to others; rare inflammation risk |
| Aflibercept (2mg) | VEGF-A, VEGF-B, PlGF | +17.3 to +17.5 | 300–450 μm | Every 4–8 weeks | Robust safety record |
| Ranibizumab | VEGF-A | +17 to +19 | 300–450 μm | Every 4 weeks | Robust safety record; low inflammation risk |
| Bevacizumab | VEGF-A | Non-inferior to aflibercept | Similar to aflibercept | Every 4 weeks | Robust safety record; lowest cost |
These insights help refine treatment choices for personalized RVO care.
Conclusion: The Future of RVO Treatment and Local Care Options
Advancements in RVO treatment are paving the way for better outcomes. Treatments like Faricimab, with its dual-pathway inhibition and extended dosing schedules, are reducing the need for frequent injections. Meanwhile, promising developments in gene therapy aim to further ease the burden of ongoing care for patients. These strides are helping to create a more tailored and patient-focused approach to managing RVO.
Personalized care is becoming increasingly possible with tools such as OCT biomarkers and widefield angiography, which allow doctors to customize treatments – whether through anti-VEGF injections or steroid implants. Acting quickly is crucial, as delays of even three to six months can result in irreversible vision loss.
"I think our most significant issue is one of patient adherence… Patients who need to return for careful follow‑up and regular treatment tend to become non‑compliant over time due to work and other personal commitments." – Michael Singer, MD, Clinical Professor of Ophthalmology
While treatment options continue to evolve, having local care options remains essential for effective management. For those in the Cheyenne area, the Cheyenne Eye Clinic and Surgery Center provides access to advanced diagnostic tools and the latest FDA-approved treatments, such as intravitreal anti-VEGF injections and steroid implants. By offering comprehensive retinal care close to home, they help patients maintain the consistent follow-up needed to protect their vision – without the added stress of traveling long distances.
With thousands of people in the U.S. affected by RVO each year, the combination of cutting-edge therapies and accessible local care brings new hope to those battling this vision-threatening condition.
FAQs
How do I know if faricimab is right for my RVO?
When considering treatment for retinal vein occlusion (RVO), it’s important to consult your ophthalmologist about whether faricimab might be a good option for you. They’ll evaluate aspects like your medical history, current condition, and the latest research. Since faricimab is a relatively new treatment being studied for RVO, a thorough discussion with your doctor is crucial to determine if it aligns with your needs.
How often will I need eye injections with newer treatments?
New treatments for retinal vein occlusion are reducing the need for frequent injections. Depending on the specific therapy, patients might only need injections every two to three months or, in some cases, as little as twice a year. For instance, certain advanced options for macular edema have greatly cut down the need for monthly injections.
When should I switch from anti-VEGF to a steroid implant?
If anti-VEGF therapy isn’t delivering the desired results or seems ineffective over time, it might be worth considering a steroid implant. Research indicates that making the switch early could lead to better results, particularly in cases like diabetic macular edema or retinal vein occlusion. It’s important to discuss this option with your eye care specialist to figure out the right timing and approach for your situation.
